Draw the Structures of XeF4, XeF6, and XeOF4 | Class 12 Chemistry Ch 7 Q 7.15
Understanding the structures of xenon compounds is fascinating because it challenges the old belief that noble gases are completely inert. These three compounds—XeF4, XeF6, and XeOF4—showcase how xenon can form stable compounds with different geometries based on VSEPR theory and hybridization.
EXAM TIP: Looking for the Perfect Answer to Write in Your Exam?
Skip the long explanation and get straight to the exam-ready answer format that will help you score full marks! We’ve prepared the perfect answer structure with proper marking scheme breakdown for this 5-mark question.
📝 Jump to Exam-Ready Answer (5 Marks) →📌 Quick Answer
XeF4 has square planar geometry (sp3d2 hybridization, 2 lone pairs). XeF6 has distorted octahedral geometry (sp3d3 hybridization, 1 lone pair). XeOF4 has square pyramidal geometry (sp3d2 hybridization, 1 lone pair with oxygen at apex). All three demonstrate xenon’s ability to expand its octet using vacant d-orbitals.
🔬 Understanding Xenon Compounds
Before we dive into the structures, it’s important to understand why xenon forms compounds at all. Xenon is a noble gas with a complete octet, but it has vacant d-orbitals in its valence shell. When xenon reacts with highly electronegative elements like fluorine and oxygen, electrons from filled p-orbitals can be promoted to these vacant d-orbitals, allowing xenon to form bonds. This is called expansion of octet or hypervalency.
The geometry of these compounds is determined by VSEPR (Valence Shell Electron Pair Repulsion) theory, which states that electron pairs around a central atom arrange themselves to minimize repulsion. Both bonding pairs and lone pairs must be considered when determining molecular shape.
📐 Structure 1: XeF4 (Xenon Tetrafluoride)
⚛️ Molecular Details
sp3d2
Square Planar
2 (axial positions)
AX4E2
🎨 Visual Structure
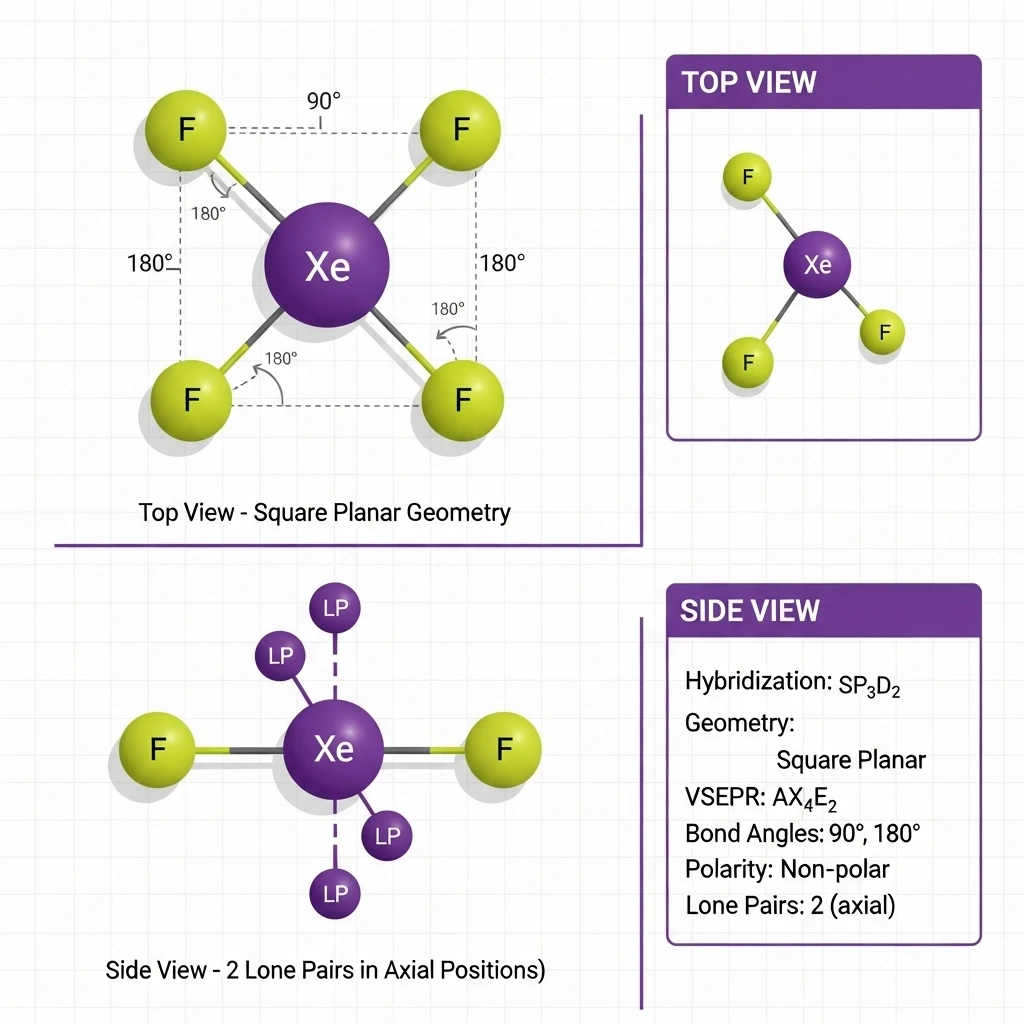
Top View (Square Planar):
F
|
|
F -------- Xe -------- F
|
|
F
All four F atoms lie in the same plane
Bond angles: F-Xe-F = 90° and 180°
Side View (showing lone pairs):
⚫ (Lone pair above)
|
|
F -------- Xe -------- F
|
|
⚫ (Lone pair below)
Two lone pairs occupy axial positions
(above and below the square plane)
📊 Hybridization Process
Ground state of Xe: [Kr] 5s2 5p6
Excited state: Two electrons from 5p are promoted to 5d orbitals → [Kr] 5s2 5p4 5d2
Hybridization: One 5s + three 5p + two 5d orbitals mix to form six sp3d2 hybrid orbitals arranged octahedrally.
Orbital usage: Four orbitals form sigma bonds with four F atoms (in equatorial plane), and two orbitals contain lone pairs (in axial positions).
📐 Structure 2: XeF6 (Xenon Hexafluoride)
⚛️ Molecular Details
sp3d3
Distorted Octahedral
1 (stereochemically active)
AX6E1
🎨 Visual Structure
Distorted Octahedral Structure:
F
|
| (slightly bent)
F -------- Xe -------- F
/|\
/ | \
/ | \
F F F
The lone pair causes distortion
Bond angles deviate from perfect 90°
3D Representation:
F (axial)
↑
|
F ← Xe → F (equatorial, distorted)
↙ ↓ ↘
F F F
⚫ Lone pair occupies space
(causes asymmetry and distortion)
📊 Hybridization Process
Ground state of Xe: [Kr] 5s2 5p6
Excited state: Electrons promoted → [Kr] 5s2 5p3 5d3
Hybridization: One 5s + three 5p + three 5d orbitals mix to form seven sp3d3 hybrid orbitals arranged in pentagonal bipyramidal geometry.
Orbital usage: Six orbitals form sigma bonds with six F atoms, and one orbital contains the lone pair.
📐 Structure 3: XeOF4 (Xenon Oxytetrafluoride)
⚛️ Molecular Details
sp3d2
Square Pyramidal
1 (below square plane)
AX5E1
🎨 Visual Structure
Square Pyramidal Structure:
O (apex)
║
║ (double bond)
Xe
/|\\
/ | \\
/ | \\
F F F
|
F
Oxygen at apex (top of pyramid)
Four F atoms form square base
Lone pair below the base
Top View (looking down from O):
F
|
|
F -------- Xe -------- F
|
|
F
⚫ Lone pair (below plane, not visible)
Side View:
O (at top)
║
║
F ------- Xe ------- F
|
F
|
⚫ (Lone pair at bottom)
📊 Hybridization Process
Ground state of Xe: [Kr] 5s2 5p6
Excited state: [Kr] 5s2 5p4 5d2
Hybridization: One 5s + three 5p + two 5d orbitals mix to form six sp3d2 hybrid orbitals arranged octahedrally.
Orbital usage: One orbital forms a double bond with oxygen (at apex), four orbitals form sigma bonds with four F atoms (square base), and one orbital contains the lone pair (opposite to oxygen).
📊 Comparison Table: All Three Structures
| Property | XeF4 | XeF6 | XeOF4 |
|---|---|---|---|
| Hybridization | sp3d2 | sp3d3 | sp3d2 |
| Molecular Geometry | Square Planar | Distorted Octahedral | Square Pyramidal |
| VSEPR Notation | AX4E2 | AX6E1 | AX5E1 |
| Number of Lone Pairs | 2 | 1 | 1 |
| Bond Angles | 90°, 180° | ~90° (distorted) | ~90° |
| Polarity | Non-polar | Polar | Polar |
| Physical State | Colorless crystals | Pale yellow solid | Colorless liquid |
📝 How to Write This Answer in Your Exam (5 Marks)
🎯 Marking Scheme Breakdown
✅ Part (a) XeF4: 1.5 marks
- Structure diagram with labels (0.5 mark)
- Hybridization and geometry (0.5 mark)
- Lone pair positions (0.5 mark)
✅ Part (b) XeF6: 1.5 marks
- Structure diagram with labels (0.5 mark)
- Hybridization and geometry (0.5 mark)
- Mention of distortion (0.5 mark)
✅ Part (c) XeOF4: 2 marks
- Structure diagram with labels (0.75 mark)
- Hybridization and geometry (0.75 mark)
- Position of oxygen and lone pair (0.5 mark)
✍️ Model Answer Format
(a) XeF4 (Xenon Tetrafluoride):
Hybridization: sp3d2
Geometry: Square planar
Lone pairs: 2 (in axial positions above and below the plane)
[Draw the structure showing Xe at center with 4 F atoms in square planar arrangement and indicate 2 lone pairs in axial positions]
(b) XeF6 (Xenon Hexafluoride):
Hybridization: sp3d3
Geometry: Distorted octahedral
Lone pairs: 1 (causes distortion from perfect octahedral geometry)
[Draw the structure showing Xe at center with 6 F atoms in distorted octahedral arrangement and indicate 1 lone pair causing the distortion]
(c) XeOF4 (Xenon Oxytetrafluoride):
Hybridization: sp3d2
Geometry: Square pyramidal
Lone pairs: 1 (below the square base, opposite to oxygen)
Special feature: Oxygen occupies the apex position with a double bond to Xe
[Draw the structure showing Xe at center, O at apex with double bond, 4 F atoms forming square base, and indicate 1 lone pair below]
✅ Detailed Step-by-Step Solution
🔍 Approach to Drawing Xenon Compound Structures
Xenon has 8 valence electrons. Count the electrons needed for bonding with F and O atoms.
Use the formula: Hybridization = ½(V + M – C + A), where V = valence electrons of central atom, M = monovalent atoms, C = cationic charge, A = anionic charge.
Count bonding pairs and lone pairs. Use VSEPR notation (AXnEm) to determine molecular geometry.
Place the central atom (Xe) and arrange bonded atoms and lone pairs according to the geometry. Label all atoms and indicate lone pairs clearly.
📐 Detailed Calculations
For XeF4:
Hybridization number = ½(8 + 4 – 0 + 0) = ½(12) = 6
This means 6 hybrid orbitals → sp3d2 hybridization
Bonding pairs = 4, Lone pairs = 2
VSEPR: AX4E2 → Square planar geometry
For XeF6:
Hybridization number = ½(8 + 6 – 0 + 0) = ½(14) = 7
This means 7 hybrid orbitals → sp3d3 hybridization
Bonding pairs = 6, Lone pairs = 1
VSEPR: AX6E1 → Distorted octahedral geometry
For XeOF4:
Hybridization number = ½(8 + 4 – 0 + 0) = ½(12) = 6
(Oxygen is divalent, counted as part of bonding arrangement)
This means 6 hybrid orbitals → sp3d2 hybridization
Bonding pairs = 5 (1 double bond with O + 4 single bonds with F), Lone pairs = 1
VSEPR: AX5E1 → Square pyramidal geometry
❓ Frequently Asked Questions (FAQs)
✍️ Written by Dr. Irfan Mansuri
Senior Science Educator & Researcher
Independent Educational Consultant • India
Dr. Irfan Mansuri brings 25 years of extensive experience in science education, specializing in Physics and Chemistry. With a doctoral degree and a passion for making science engaging, he has mentored countless students to achieve excellence in their board examinations. His teaching methodology emphasizes conceptual clarity, practical applications, and exam-oriented strategies that help students not just memorize, but truly understand scientific principles.