The p-Block Elements: Complete Guide for Class 12 Chemistry | Groups 13-18 Summary
Welcome to the most comprehensive guide on p-block elements! If you’re preparing for CBSE Class 12 boards, JEE, or NEET, this complete summary covers everything you need to know about Groups 13 through 18 of the periodic table. Let’s master these elements together! ⚗️
EXAM ALERT: This Chapter is a Goldmine for Board & Competitive Exams!
p-Block elements contribute 15-20% of your Chemistry paper! This summary covers all groups with trends, reactions, and important compounds. Master this, and you’ll ace questions worth 15-20 marks in boards and multiple questions in JEE/NEET!
🎯 Jump to Exam Strategy & Important Questions →⚡ Quick Overview: What Are p-Block Elements?
p-Block elements are those elements in which the last electron enters the p-orbital. They occupy Groups 13-18 of the periodic table and include metals, metalloids, and non-metals. These elements show diverse properties and form the basis of numerous important compounds in chemistry and everyday life!
📊 Complete Groups Overview
| Group | Name | Elements | Configuration | Key Characteristics |
|---|---|---|---|---|
| 13 | Boron Family | B, Al, Ga, In, Tl | \(ns^2 np^1\) | Metallic character increases down the group; +3 oxidation state common |
| 14 | Carbon Family | C, Si, Ge, Sn, Pb | \(ns^2 np^2\) | Shows catenation; +4 and +2 oxidation states; allotropy common |
| 15 | Nitrogen Family | N, P, As, Sb, Bi | \(ns^2 np^3\) | Diatomic N₂; P₄ tetrahedral; oxidation states -3 to +5 |
| 16 | Oxygen Family (Chalcogens) | O, S, Se, Te, Po | \(ns^2 np^4\) | Oxygen is diatomic; sulfur shows allotropy; -2 to +6 oxidation states |
| 17 | Halogen Family | F, Cl, Br, I, At | \(ns^2 np^5\) | Most reactive non-metals; strong oxidizing agents; -1 to +7 oxidation states |
| 18 | Noble Gases | He, Ne, Ar, Kr, Xe, Rn | \(ns^2 np^6\) | Chemically inert; complete octet; Xe forms compounds |
📈 Important Periodic Trends in p-Block
🔹 1. Atomic and Ionic Radii
Down the group: Atomic and ionic radii increase due to addition of new electron shells.
Across the period: Atomic radius decreases from Group 13 to 18 due to increasing nuclear charge.
⚠️ Exception Alert: In Group 13, Gallium has a smaller radius than Aluminum due to poor shielding by d-electrons (d-block contraction). Similarly, in Group 15, the radii of As, Sb, and Bi are similar due to d and f-block contractions.
🔹 2. Ionization Enthalpy
Down the group: Ionization enthalpy decreases due to increasing atomic size and shielding effect.
Across the period: Ionization enthalpy increases from Group 13 to 18 due to decreasing atomic size.
💡 Key Point: Group 18 (noble gases) have the highest ionization enthalpy in each period due to stable electronic configuration. Group 13 elements have lower ionization enthalpy than Group 2 due to penetration effect.
🔹 3. Electronegativity
Down the group: Electronegativity decreases due to increasing atomic size.
Across the period: Electronegativity increases from Group 13 to 17. Group 18 elements don’t form bonds, so electronegativity is not defined.
Most electronegative element: Fluorine (F) with electronegativity value of 4.0 on the Pauling scale.
🔹 4. Metallic Character
Down the group: Metallic character increases as ionization enthalpy decreases.
Across the period: Metallic character decreases from Group 13 to 17.
| Group | Metals | Metalloids | Non-metals |
|---|---|---|---|
| 13 | Al, Ga, In, Tl | B | — |
| 14 | Sn, Pb | Si, Ge | C |
| 15 | Sb, Bi | As | N, P |
| 16 | Po | Te | O, S, Se |
| 17 | — | At (debated) | F, Cl, Br, I |
⚗️ Group-Wise Detailed Summary
13 Group 13: Boron Family
Elements: Boron (B), Aluminum (Al), Gallium (Ga), Indium (In), Thallium (Tl)
Electronic Configuration: \(ns^2 np^1\)
🔸 Important Properties:
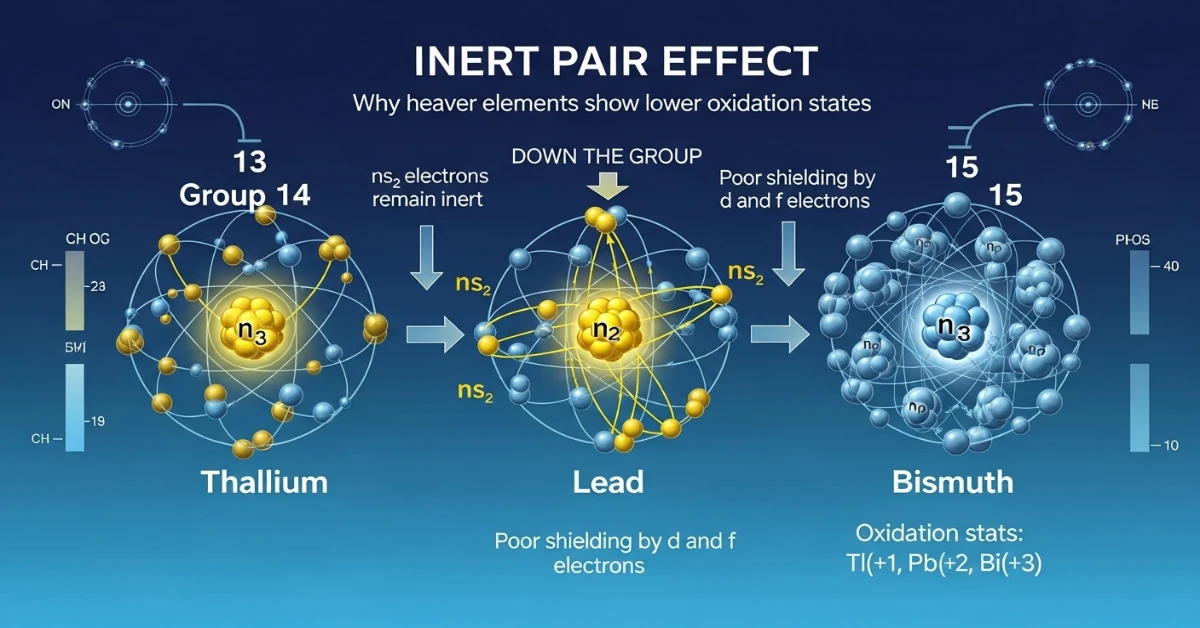
- Oxidation States: Primarily +3, but Tl also shows +1 (inert pair effect)
- Boron: Only non-metal; forms covalent compounds; electron deficient (only 6 electrons in valence shell)
- Aluminum: Most abundant metal in Earth’s crust; amphoteric nature
- Inert Pair Effect: Increases down the group (most prominent in Tl)
🔸 Important Compounds:
1. Borax (Na₂B₄O₇·10H₂O)
Used in glass manufacturing, as antiseptic, and in detergents
2. Boric Acid (H₃BO₃)
Weak monobasic Lewis acid; used as antiseptic
3. Aluminum Oxide (Al₂O₃)
Amphoteric; used in extraction of aluminum
14 Group 14: Carbon Family
Elements: Carbon (C), Silicon (Si), Germanium (Ge), Tin (Sn), Lead (Pb)
Electronic Configuration: \(ns^2 np^2\)
🔸 Important Properties:
- Oxidation States: +4 and +2 (inert pair effect increases down the group)
- Catenation: C > Si >> Ge > Sn > Pb (carbon shows maximum catenation)
- Allotropy: Carbon (diamond, graphite, fullerenes), Tin (α-tin, β-tin)
- Carbon: Unique ability to form multiple bonds (C=C, C≡C)
🔸 Important Compounds:
1. Carbon Monoxide (CO)
Neutral oxide; highly toxic; forms carboxyhemoglobin
2. Carbon Dioxide (CO₂)
Acidic oxide; greenhouse gas; used in fire extinguishers
3. Silicon Dioxide (SiO₂)
Acidic oxide; used in glass, ceramics, and semiconductors
15 Group 15: Nitrogen Family (Pnictogens)
Elements: Nitrogen (N), Phosphorus (P), Arsenic (As), Antimony (Sb), Bismuth (Bi)
Electronic Configuration: \(ns^2 np^3\)
🔸 Important Properties:
- Oxidation States: -3 to +5 (most common: -3, +3, +5)
- Nitrogen: Exists as diatomic N₂ with triple bond (very stable)
- Phosphorus: Exists as P₄ (tetrahedral); shows allotropy (white, red, black)
- Metallic Character: Increases from N to Bi (Bi is metallic)
🔸 Why NH₃ forms H-bonds but PH₃ doesn’t?
Nitrogen is highly electronegative and small in size, creating strong N-H bonds with high polarity. This allows NH₃ to form strong hydrogen bonds. Phosphorus is larger and less electronegative, so P-H bonds are less polar and cannot form effective hydrogen bonds.
🔸 Important Compounds:
1. Ammonia (NH₃)
Basic gas; Haber process; used in fertilizers
2. Nitric Acid (HNO₃)
Strong oxidizing agent; prepared by Ostwald process
3. Phosphoric Acid (H₃PO₄)
Tribasic acid; used in fertilizers and soft drinks
16 Group 16: Oxygen Family (Chalcogens)
Elements: Oxygen (O), Sulfur (S), Selenium (Se), Tellurium (Te), Polonium (Po)
Electronic Configuration: \(ns^2 np^4\)
🔸 Important Properties:
- Oxidation States: -2 to +6 (most common: -2, +4, +6)
- Oxygen: Exists as O₂ (diatomic); shows allotropy (O₂, O₃)
- Sulfur: Shows allotropy (rhombic, monoclinic); exists as S₈
- Why H₂O is liquid but H₂S is gas? Strong H-bonding in H₂O increases boiling point
🔸 Important Compounds:
1. Ozone (O₃)
Powerful oxidizing agent; protects from UV radiation
2. Sulfuric Acid (H₂SO₄)
King of chemicals; Contact process; strong dehydrating agent
3. Sulfur Dioxide (SO₂)
Acidic gas; bleaching agent; causes acid rain
17 Group 17: Halogen Family
Elements: Fluorine (F), Chlorine (Cl), Bromine (Br), Iodine (I), Astatine (At)
Electronic Configuration: \(ns^2 np^5\)
🔸 Important Properties:
- Oxidation States: -1 to +7 (F shows only -1)
- Most Reactive Non-metals: Reactivity decreases F > Cl > Br > I
- Strong Oxidizing Agents: Oxidizing power decreases down the group
- Physical State: F₂, Cl₂ (gases); Br₂ (liquid); I₂ (solid)
- Why F forms only one oxoacid? High electronegativity and small size
🔸 Important Compounds:
1. Hydrochloric Acid (HCl)
Strong acid; used in labs and industry
2. Chlorine (Cl₂)
Used in water purification and bleaching
3. Interhalogen Compounds
ClF₃, BrF₅, IF₇ – more reactive than parent halogens
18 Group 18: Noble Gases
Elements: Helium (He), Neon (Ne), Argon (Ar), Krypton (Kr), Xenon (Xe), Radon (Rn)
Electronic Configuration: \(ns^2 np^6\) (except He: \(1s^2\))
🔸 Important Properties:
- Chemically Inert: Complete octet (stable electronic configuration)
- Very Low Boiling Points: Weak van der Waals forces
- Monoatomic Gases: Exist as single atoms
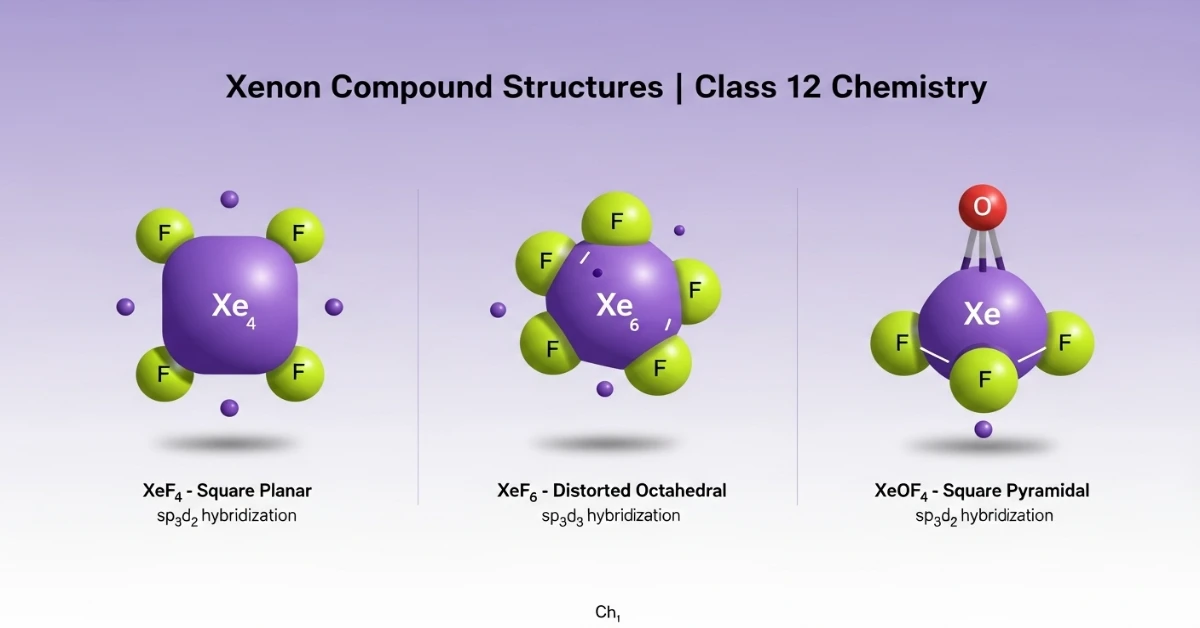
- Xenon Compounds: XeF₂, XeF₄, XeF₆, XeO₃, XeOF₄
- Why Xe forms compounds but He doesn’t? Larger size, lower ionization energy
🎯 Exam Strategy & Important Topics
📝 High-Weightage Topics for Board Exams
- Trends in physical and chemical properties (3-5 marks)
- Anomalous behavior of first elements (2-3 marks)
- Inert pair effect (2-3 marks)
- Preparation and properties of important compounds (3-5 marks)
- Oxoacids of halogens and Group 15, 16 (3-5 marks)
- Noble gas compounds (2-3 marks)
- Comparison questions (Why X but not Y?) (2-3 marks)
💡 Memory Tricks for p-Block Elements
Group 13 (B, Al, Ga, In, Tl):
“BAG of INTeLligence” – B, Al, Ga, In, Tl
Group 15 (N, P, As, Sb, Bi):
“Naughty Pupils Are Studying Biology” – N, P, As, Sb, Bi
Group 17 (F, Cl, Br, I, At):
“Fools Carry Bricks In Attics” – F, Cl, Br, I, At
Oxidizing Power of Halogens:
“F > Cl > Br > I” (Remember: Smaller = Stronger oxidizer)
⚠️ Common Mistakes to Avoid
- Mistake: Confusing oxidation states of different groups
Solution: Make a separate chart for each group’s oxidation states - Mistake: Not explaining “why” in comparison questions
Solution: Always give reasons (size, electronegativity, bonding, etc.) - Mistake: Forgetting exceptions (like Ga radius < Al radius)
Solution: Highlight all exceptions in your notes - Mistake: Writing wrong chemical formulas
Solution: Practice writing formulas daily (especially oxoacids)
❓ Frequently Asked Questions (FAQs)
1. Why does the metallic character increase down the group in p-block elements?
As we move down the group, atomic size increases and ionization enthalpy decreases. This makes it easier for atoms to lose electrons, which is a characteristic property of metals. Therefore, metallic character increases from top to bottom in p-block groups. For example, in Group 14, carbon is a non-metal while lead is a metal.
2. What is the inert pair effect and in which groups is it most prominent?
The inert pair effect is the tendency of the \(ns^2\) electron pair to remain non-bonding in heavier elements of groups 13-16. This happens because the s-electrons are held tightly by the nucleus due to poor shielding by d and f electrons. It’s most prominent in Groups 13 (Tl shows +1), 14 (Pb shows +2), and 15 (Bi shows +3) instead of their maximum oxidation states.
3. Why is nitrogen a gas while phosphorus is a solid at room temperature?
Nitrogen exists as diatomic N₂ molecules with a very stable triple bond (N≡N). These small molecules have weak van der Waals forces between them, making nitrogen a gas. Phosphorus exists as P₄ tetrahedral molecules, which are larger and have stronger intermolecular forces, making it a solid at room temperature.
4. Why does fluorine form only one oxoacid (HOF) while other halogens form multiple oxoacids?
Fluorine is the most electronegative element and has a very small atomic size. It cannot accommodate multiple oxygen atoms around it, and it doesn’t have d-orbitals for expansion of its octet. Therefore, it forms only one oxoacid (HOF – hypofluorous acid). Other halogens have d-orbitals and can form multiple oxoacids with different oxidation states.
5. Why does H₂O have a higher boiling point than H₂S despite H₂S being heavier?
Water (H₂O) forms strong hydrogen bonds due to the high electronegativity and small size of oxygen. These hydrogen bonds require significant energy to break, resulting in a high boiling point (100°C). H₂S has weaker van der Waals forces because sulfur is larger and less electronegative, so it cannot form effective hydrogen bonds. Therefore, H₂S has a much lower boiling point (-60°C) and exists as a gas at room temperature.
6. Why can xenon form compounds but helium cannot?
Xenon has a larger atomic size and lower ionization enthalpy compared to helium. The valence electrons in xenon are farther from the nucleus and less tightly held, making it possible for highly electronegative elements like fluorine and oxygen to remove or share these electrons. Helium has a very small size and extremely high ionization enthalpy, making it virtually impossible to form compounds under normal conditions.
7. What is the difference between rhombic and monoclinic sulfur?
Both are allotropes of sulfur consisting of S₈ rings. Rhombic sulfur (α-sulfur) is the most stable form at room temperature, has a yellow color, and crystallizes in octahedral shapes. Monoclinic sulfur (β-sulfur) is stable above 96°C, has a pale yellow color, and crystallizes in needle-like shapes. They can interconvert depending on temperature.
8. Why is carbon unique in forming multiple bonds while silicon cannot?
Carbon is small in size, which allows effective sideways overlap of p-orbitals to form strong π-bonds (C=C, C≡C). Silicon is much larger, and the p-orbitals are too far apart for effective π-overlap. Therefore, silicon prefers to form single bonds (Si-Si) rather than multiple bonds. This is why carbon forms countless organic compounds with double and triple bonds, while silicon chemistry is dominated by single bonds.
9. Why is boric acid considered a weak acid despite having three OH groups?
Boric acid (H₃BO₃) is a weak monobasic Lewis acid, not a typical Brønsted acid. It doesn’t donate H⁺ ions directly. Instead, it accepts OH⁻ ions from water to form [B(OH)₄]⁻, releasing H⁺ ions in the process: H₃BO₃ + H₂O → [B(OH)₄]⁻ + H⁺. This unique mechanism makes it a weak acid despite having three hydroxyl groups.
10. What is the order of acidic strength of halogen acids and why?
The acidic strength order is: HI > HBr > HCl > HF. As we move down the group, the bond length increases and bond strength decreases, making it easier to release H⁺ ions. HF is the weakest despite fluorine being most electronegative because it forms strong hydrogen bonds, making H⁺ release difficult. HI is the strongest acid because the H-I bond is longest and weakest.
🔬 Important Reactions Summary
⚗️ Group 15 – Key Reactions
1. Haber Process (Ammonia Production):
N₂ + 3H₂ ⇌ 2NH₃ (200 atm, 700 K, Fe catalyst)
2. Ostwald Process (Nitric Acid Production):
4NH₃ + 5O₂ → 4NO + 6H₂O
2NO + O₂ → 2NO₂
4NO₂ + O₂ + 2H₂O → 4HNO₃
3. Reaction with Metals:
3Mg + N₂ → Mg₃N₂ (magnesium nitride)
⚗️ Group 16 – Key Reactions
1. Contact Process (Sulfuric Acid Production):
S + O₂ → SO₂
2SO₂ + O₂ ⇌ 2SO₃ (V₂O₅ catalyst)
SO₃ + H₂SO₄ → H₂S₂O₇ (then diluted to H₂SO₄)
2. Ozone Formation:
3O₂ → 2O₃ (UV radiation or silent electric discharge)
3. H₂O₂ as Oxidizing Agent:
H₂O₂ + 2H⁺ + 2e⁻ → 2H₂O
⚗️ Group 17 – Key Reactions
1. Bleaching Action of Cl₂:
Cl₂ + H₂O → HCl + HOCl (HOCl releases nascent oxygen)
2. Reaction with Alkali:
Cl₂ + 2NaOH → NaCl + NaOCl + H₂O (cold, dilute)
3. Interhalogen Formation:
Cl₂ + F₂ → 2ClF (chlorine monofluoride)
⚗️ Group 18 – Key Reactions
1. Xenon Fluorides Formation:
Xe + F₂ → XeF₂ (1:1 ratio, 673 K)
Xe + 2F₂ → XeF₄ (1:5 ratio, 873 K)
Xe + 3F₂ → XeF₆ (1:20 ratio, 573 K, high pressure)
2. Hydrolysis of XeF₆:
XeF₆ + 3H₂O → XeO₃ + 6HF
📚 Quick Revision Table
| Property | Down the Group | Across the Period (13→18) | Reason |
|---|---|---|---|
| Atomic Radius | Increases ↑ | Decreases → | New shell added / Nuclear charge increases |
| Ionization Energy | Decreases ↓ | Increases → | Atomic size / Effective nuclear charge |
| Electronegativity | Decreases ↓ | Increases → | Atomic size / Nuclear attraction |
| Metallic Character | Increases ↑ | Decreases → | Ease of losing electrons |
| Oxidizing Power | Decreases ↓ | Increases → | Electron affinity / Electronegativity |
| Boiling Point | Increases ↑ | Varies | Van der Waals forces / Molecular size |
🎓 Practice Questions for Board Exams
✍️ 2-3 Mark Questions
- Why does nitrogen show catenation properties less than phosphorus?
- Explain why BiH₃ is the strongest reducing agent amongst all the hydrides of Group 15 elements.
- Why are pentahalides more covalent than trihalides?
- Arrange H₂O, H₂S, H₂Se, H₂Te in order of their acidic strength and justify.
- Why is ICl more reactive than I₂?
📖 3-5 Mark Questions
- Explain the manufacture of sulfuric acid by Contact process with chemical equations.
- Draw the structures of (a) XeF₄ (b) XeF₆ (c) XeOF₄
- Compare the properties of ammonia and phosphine.
- Describe the trends in physical and chemical properties of Group 17 elements.
- Explain why H₂O₂ acts as both oxidizing and reducing agent with examples.
💯 Final Tips for Scoring Maximum Marks
🏆 Expert Tips from Dr. Irfan Mansuri
- Make Comparison Charts: Create tables comparing properties of elements within each group and across periods
- Practice Chemical Equations: Write all important reactions daily – especially preparation methods of compounds
- Understand “Why” Questions: Don’t just memorize – understand the reasoning behind trends and exceptions
- Draw Structures: Practice drawing structures of important compounds like XeF₄, H₂SO₄, HNO₃, etc.
- Focus on Exceptions: Pay special attention to anomalous behavior and exceptions in trends
- Solve Previous Year Papers: Identify frequently asked questions and practice them thoroughly
- Use Mnemonics: Create memory tricks for remembering element sequences and properties
- Link to Real Life: Connect concepts to real-life applications for better retention
🎯 Remember: p-Block Elements = 15-20% of Your Chemistry Paper!
Master these concepts, practice regularly, and you’ll confidently score full marks in this chapter. Focus on understanding trends, exceptions, and important reactions. Good luck with your preparation! 🚀
👨🏫 About the Author
Dr. Irfan Mansuri is a Chemistry expert with over 25 years of teaching experience. He has helped thousands of students excel in CBSE Board exams, JEE, and NEET. His teaching methodology focuses on conceptual clarity, practical applications, and exam-oriented preparation. This comprehensive guide is designed to help Class 12 students master p-block elements and score maximum marks in their Chemistry exams.
📌 Key Takeaways
- p-Block elements include Groups 13-18 with diverse properties
- Metallic character increases down the group, decreases across period
- Inert pair effect is prominent in heavier elements of Groups 13-15
- Fluorine is most electronegative; forms only one oxoacid
- Noble gases are chemically inert; only Xe forms stable compounds
- Important industrial processes: Haber, Ostwald, Contact process
- Hydrogen bonding explains anomalous properties of H₂O, NH₃, HF
- Allotropy is common in Groups 14, 15, and 16
🌟 Master p-Block Elements & Ace Your Chemistry Exam! 🌟
Study this guide thoroughly, practice regularly, and success will be yours! 📚✨